|
Medline for chromium metabolism AND bacteria Chromate influx is known to occur via sulfate uptake systems in some microorganisms Chromium resistance has also been described in fungi and algae, and some of these organismsĬan immobilize soluble chromium via bioaccumulation and biosorption (reviewed by Cervantes et al, 2001). Chromium(VI) reduction is likely a detoxification mechanism, butīacterial chromium resistance is also mediated by non-reductive plasmid-based systems that appear to exclude chromium (as chromate ions) from Cytochromes of type c and b have been implicated in chromium reduction,īut the mechanisms of chromium bioreduction have not been completely determined (reviewed by Lovley, 1993). Microbial chromium(VI) reduction has been observed most often under aerobic conditions, but can also occur under anaerobic conditions (reviewed by Wang and Shen, 1995). However, no beneficial function of chromium has been Involve reduction of chromium(VI) to less toxic and less mobile forms suchĬhromium(III) is an essential trace element in humans, where it enhances In this context, most studies on prokaryotes and chromium Chromium(VI) is a known human carcinogen. However, is largely substitution inert and serves poorly The latter metals often serve as enzyme cofactors. Neighbors in the Periodic Table such as manganese and iron. With biological systems are very different from that of its Wild electron configurations also mean you have a good chance at a variety of valence numbers.Chromium is a first row transition metal, but its interactions Potassium, chromium, and copper are the only elements in the fourth period that have one electron in the outermost shell. While vanadium (to the left) added one electron to its third shell, chromium adds its electron to the third shell and has one electron from the fourth shell bumped down. Being a transition metal, the electron configuration is unique. The fourth row of the periodic table has transition metals ranging from scandium (21) to zinc (30).Ĭhromium is number twenty-four on the periodic table and located in the same column with molybdenum and tungsten. Remember that the first eight were placed during our trip through the third period/row. 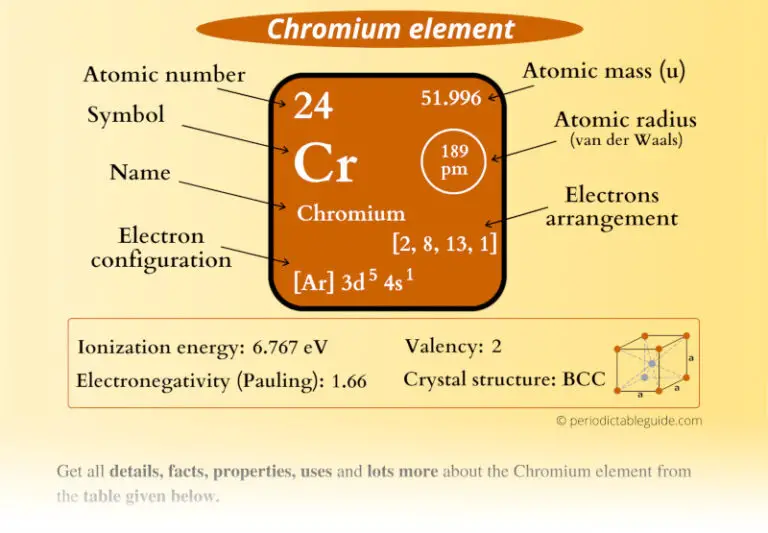
For the fourth period/row, all of these electrons build the third shell to a maximum of 18 electrons. This element is one of the transition elements that doesn't place the additional electrons in the outer shell, but in the one underneath. So remember when you look at our breakdown that the electrons aren't always in a nice neat order as shown here. They are found in clouds that can have different shapes that include spheres and dumbbell-like shapes. As you learn more about atomic structure, you will learn that the electrons don't stay in defined areas around the nucleus. The electrons like to be in separate shells/orbitals. In an atom, the electrons spin around the center, also called the nucleus. Each of those colored balls is an electron. If you think this is a little over your head, go back and look at the elements 1-18 that have organizations that are a little more simple. Let's take a look at the arrangements of electrons in the basic elements (left and right sides of the table) of period four and the more complex arrangements of the transition elements (in the middle of the row). You may have an easy way to know the number of electrons in a neutral atom, but the placement of those electrons gets a little more complex. Now we're working with the fourth period/row in the table of elements. 
It tells you the mass of one atom, how many pieces are inside, and where it should be placed on the periodic table. That box on the left has all of the information you need to know about one element.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
